“Mad Men” Meets Pharma
Following Turing Pharmaceuticals’ decision to raise the price of Daraprim, an anti-infection treatment used by HIV patients, from $13.50 to $750 per pill, the company’s former CEO Martin Shkreli appeared before Congress for a hearing on drug prices.1 Smirking and refusing to answer legislators’ questions by invoking the Fifth Amendment, which protects individuals from forced self-incrimination, Shkreli further cemented his status as a pharmaceutical industry villain.
Shkreli is arguably the most infamous figure among Big Pharma’s Mad Men. Big Pharma’s Mad Men refers to the individuals and organizations within the industry whose behavior is reminiscent of the cynical practices of 1950s New York City advertisers featured in the hit AMC series. The Mad Men of advertising worked on Madison Avenue and preyed on society’s insecurities to peddle consumer goods, such as cigarettes, without regard for their impact on the people consuming them.
Like the original Mad Men, Big Pharma’s Mad Men exploit lax government regulation to enrich themselves at the cost of society’s health. Their manufacturing, advertising, and sales practices have received considerable criticism, yet the industry’s financial ties to legislators have prevented significant reform and even spurred policy that favors Big Pharma over the public good. Consumers, especially those with a lower income, inevitably pay the price. Unfortunately, there’s very little citizens can do about Big Pharma except remain vigilant and support greater regulation.
Throughout this piece we will explore the roots of the pharmaceutical industry’s power and influence on American health care and politics. This information can help to educate patients and health care providers, including nurse practitioners, and support them in advocating for better care and better regulation.
The Roots of Big Pharma
Many of the largest pharmaceutical companies in the world today were founded during the 19th century and rose to prominence by holding patents to basic medicines. The early history of Bayer is illustrative: 36 years after the company’s founding in 1863, Bayer developed acetylsalicylic acid and launched the drug under its better-known name, aspirin. Despite the drug being considered an essential medicine by the World Health Organization, Bayer held the U.S. patent for aspirin for almost 20 years, granting the company a legal monopoly over its production. Aspirin’s popularity thus enriched Bayer considerably.
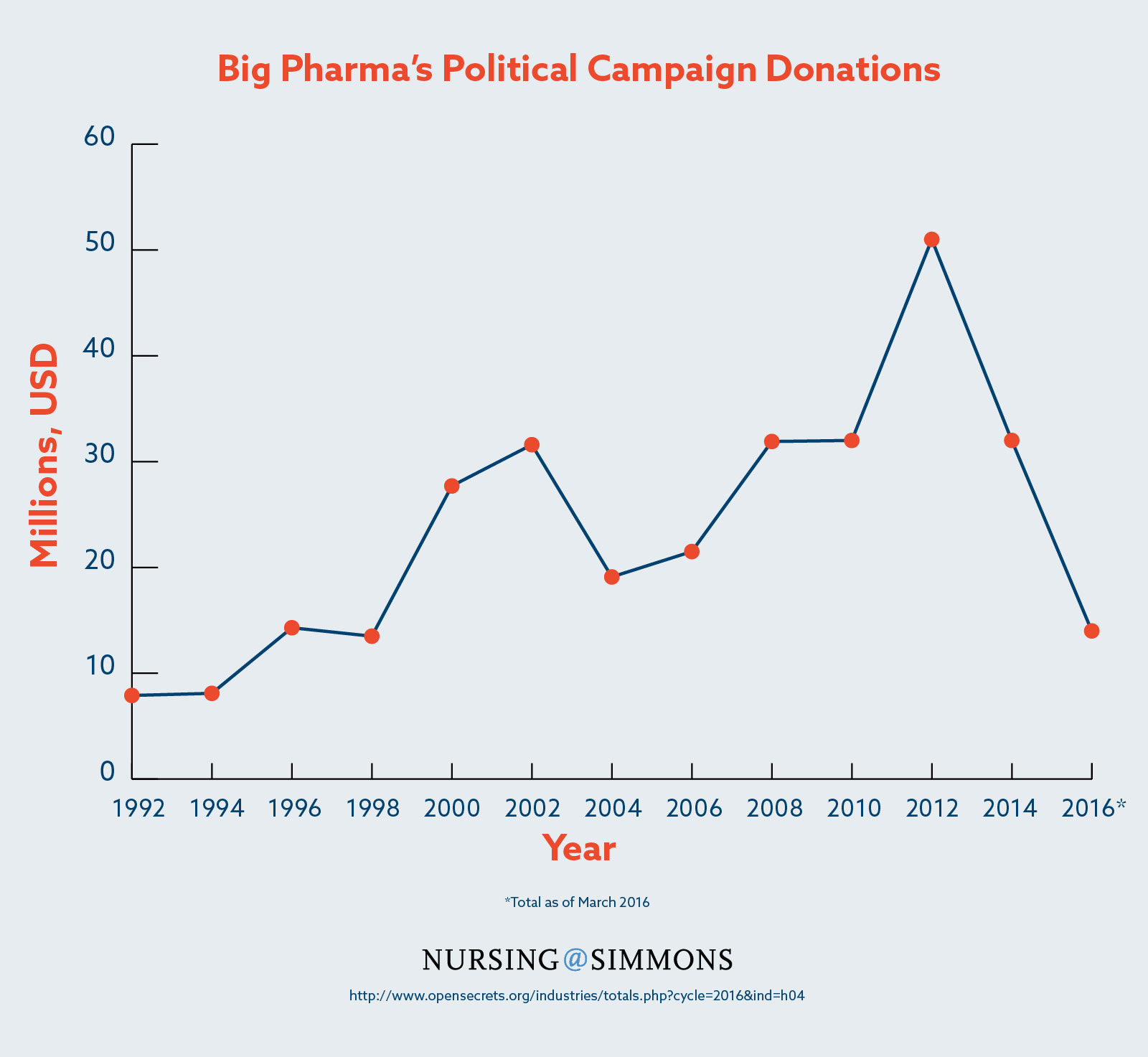
The political power of pharmaceutical companies was compounded in 1958 with the formation of the Pharmaceutical Research and Manufacturers of America (PhRMA), a trade group which would grow to become not only the largest pharmaceutical lobby in the nation but also one of the largest lobbies period. From 1998 to 2015, PhRMA spent more than $285 million on lobbying public officials, the sixth highest expenditure of any organization.2
Pharmaceuticals Today
Newly developed pharmaceuticals are eligible for 20-year patents issued by the U.S. Patent and Trademark Office, and five-year New Chemical Entity (NCE) exclusivity from the Food and Drug Administration. Ideally, these monopolies allow pharmaceutical companies to recoup the costs of research and development, but tactics intended to undercut competition and maximize profits are common. One strategy for exploiting loopholes in the system has become so routine it’s enshrined in industry jargon: evergreening. Pharmaceutical companies evergreen their products by making superficial changes that entail no beneficial innovation, yet nevertheless extend patent protections, thereby delaying the availability of more affordable generic drugs. Slightly modifying the molecular makeup of a drug is one method used to evergreen a drug.3
In a more Mad Men-esque tactic, Big Pharma has also taken to hiring sales representatives to influence physicians directly. Altogether, the pharmaceutical industry employs 90,000 sales representatives4 and spends $15 billion5 “detailing” each year. Under the guise of educational meetings, “detailing” consists of representatives visiting doctors to encourage them to prescribe particular products at the expense of competitors, cheaper generic options, and even patients’ health.6 This is accomplished through gifts ranging from branded office supplies to paid meals, cash payments for speaking engagements, and all-expenses-paid vacations thinly veiled as professional conferences.7 It has also been noted that many sales representatives are recruited from the ranks of former cheerleaders, a transparent ploy aimed at seducing physicians, who tend to be male.8 The compensation of sales representatives, 25 percent of which tends to be based on commission, encourages these questionable practices.9 This includes targeting doctors who have histories of prescribing large amounts of drugs by correlating prescription-related data obtained from hospitals with licensing information from the American Medical Association.10
Influence on Legislation
While the aforementioned abuses have been documented extensively, relatively little has been done to close known loopholes and better regulate unfair practices. The probable reasons for this include lobbying, revolving door politics, free market and small government ideologies, as well as the financial reliance regulators have on the pharmaceutical industry.
Lobbyists for the pharmaceutical industry, including PhRMA and its peers, spent nearly $3.5 billion lobbying politicians from 1998 to 2015, more than any other sector.11 This undoubtedly has an impact on the legislative decisions made by politicians. For example, the Patent Transparency and Improvements Act of 2013, which would have reduced meritless lawsuits by patent holders seeking to limit competition, was suddenly dropped by its sponsor at the behest of then Senate Majority Leader Harry Reid.12 Reid received $612,658 in campaign contributions from Big Pharma in 2012 and another $765,708 in 2014.13
The pharmaceutical industry’s methods of influencing policy take full advantage of the lax regulations on the career overlap between government and business. Known as revolving door politics, the inherent conflicts of interest borne of relationships between government officials and pharmaceutical executives become apparent when considered in particular cases. For example, in 2003 Representative Billy Tauzin, of Louisiana, helped author the Medicare Prescription Drug, Improvement, and Modernization Act (MMA), which introduced Medicare Part D, a prescription drug program. Medicare Part D requires the federal government to subsidize the cost of these prescriptions, but a provision in the MMA bars the government from negotiating prices with pharmaceutical companies. Thus, under the Medicare Part D program the Congressional Budget Office calculates that drug costs are 27 to 38 percent higher than those under Medicaid, which is able to negotiate prices with pharmaceutical companies. A few months after the MMA was signed into law, Tauzin was offered a $2 million-a-year position to join PhRMA by the president and CEO.14 While in government, Tauzin authored legislation that benefited Big Pharma, not the drug consumers he represented. He accepted the offer and left government to work for PhRMA, which highlighted a clear conflict of interest that evolved due to revolving door politics.

Rather than the FDA being obstructionist, it has been criticized for being too accommodating of the pharmaceutical industry. The FDA’s Center for Drug Evaluation and Research is responsible for regulating the safety of new medicines, but it does not conduct its own tests, relying instead on reports from the manufacturers. Furthermore, half of the FDA’s budget for regulating the industry comes from the industry itself in the form of fees paid to expedite the drug approval process. The conflict of interest is manifested in the staff of the FDA, where far more scientists are employed to review new medicines than study the long-term safety of existing ones and in the agency’s unwillingness to pull dangerous drugs from the market or limit direct-to-consumer advertising.15
Pre-Existing Regulations
The reform measures that have made it through the gauntlet described above have predictably emerged worse for the wear. Some states, like Minnesota, have passed legislation restricting gift-giving between pharmaceutical sales representatives and physicians.16 However, many of these laws contain loopholes that the industry exploits with creative pretenses, such as the aforementioned speaking fees or vacations masked as professional conferences.
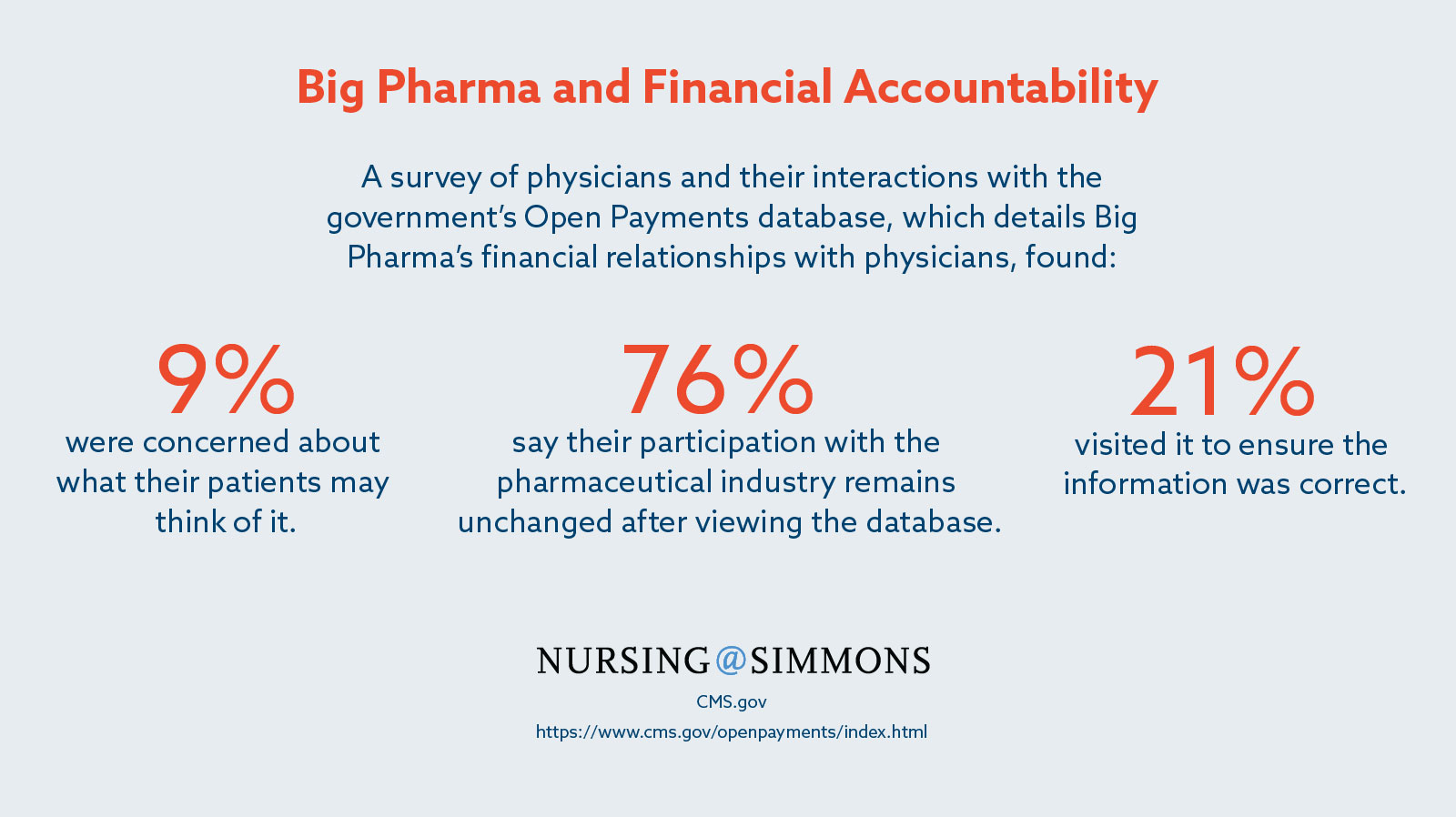
The most ambitious attempt to regulate the relationship between sales representatives and doctors is arguably Open Payments. Created by the Affordable Care Act, Open Payments is a database to which pharmaceutical manufacturers covered by Medicare, Medicaid, and the State Children’s Health Insurance Program are required to submit the details of their financial relationships with physicians. While desirable in theory, a study published in the Archives of Internal Medicine on such disclosure programs found them to have a negligible to small impact on reducing the prescription of brand-name medication in favor of generic equivalents, a reasonable measure of the influence of sales representatives on physicians. Underscoring this point, 76 percent of doctors surveyed about Open Payments claimed that their relationship with the pharmaceutical industry was unchanged following the database’s debut.17
The Victims
Victims of the pharmaceutical industry’s reach are consumers, especially those with a low income. Big Pharma’s abuse of patent laws to delay even one generic drug from reaching the market can cost consumers hundreds of millions of dollars.18 Similarly, the industry’s influence on the MMA, barring the federal government from negotiating drug prices for Medicare Part D, cost taxpayers up to $50 billion per year.19 Big Pharma’s impact on low-income consumers is perhaps the most dire. Leveraging its political influence, the pharmaceutical industry is attempting to secure protections in the Trans-Pacific Partnership trade agreement that would preserve its monopoly pricing protections abroad, effectively prioritizing its profits over the health of the impoverished, especially in developing countries.20
What Is To Be Done?
As Big Pharma has only provided token measures of self-regulation, such as its voluntary, honor-based code of conduct for sales representatives, the only recourse left to the public is vigilance and continued support for more stringent government-led reform, regardless of how unlikely its success may be.
In respect to personal health and professional standards, vigilance is key. Patients should look up their doctors on Open Payments and discuss the results with them. Health care providers can similarly research themselves and colleagues to ensure the information in the database is accurate. According to Consumer Reports, patients and health care providers alike, should also watch for these telltale signs of over-prescription as they may hint at the influence of pharmaceutical sales representatives:
- Multiple drugs prescribed without satisfactory explanation.
- Individual prescriptions for each symptom.
- Overwhelming presence of pharmaceutical industry-branded office supplies.
- Prescription of antibiotics for viral infections.
- Disinterest in nonprescription drugs and supplements.
A patient who believes their provider is over-prescribing, or a health care provider who believes the same regarding a colleague, should broach the subject directly with the individual in question. If he or she is uncooperative, a supervisor should be consulted. Many hospitals and clinics have their own policies dictating the relationships between health care providers and sales representatives or may be interested in establishing one once the issue is brought to their attention.
As far as legislative action is concerned, there are a number of current efforts that could curb the excesses of Big Pharma. The Protecting American Talent and Entrepreneurship (PATENT) Act of 2015 could reduce meritless patent lawsuits in a way similar to the Patent Transparency and Improvements Act of 2013 was intended. State legislation seems even more promising, with the California Drug Price Relief Act and the Ohio Drug Price Relief Act matching state prescription costs to the prices set by U.S. Department of Veterans Affairs. The PATENT Act is currently in the Senate, where supporters can champion it by requesting their senators cosponsor it. Similarly, supporters of the Ohio Drug Price Relief Act can pressure the state’s General Assembly to accept the ballot initiative for inclusion in the November election. The California Drug Price Relief Act, on the other hand, has already been accepted as a ballot initiative and supporters should vote for it on Election Day in November 2016.
Although the pharmaceutical industry is deeply influential in both politics and health care in the United States, patients and health care providers can still engage in the process and take steps to ensure that they receive the treatment and prescriptions that are best for them. By staying vigilant and engaging with lawmakers, people can influence their personal health and legislative policy that influences millions of people across the country and billions around the world.
1. http://www.nytimes.com/2016/02/05/business/drug-prices-valeant-martin-shkreli-congress.html?_r=0
2. https://www.opensecrets.org/lobby/top.php?indexType=s&showYear=a
3. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3680578/
4. http://www.newstatesman.com/pharmaceutical-sales-representatives-slowly-vanishing-breed
5. http://www.pewtrusts.org/en/research-and-analysis/fact-sheets/2013/11/11/persuading-the-prescribers-pharmaceutical-industry-marketing-and-its-influence-on-physicians-and-patients
6. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2464950/
7. http://www.theguardian.com/society/2008/aug/23/health.pharmaceuticals
8. http://www.nytimes.com/2005/11/28/business/gimme-an-rx-cheerleaders-pep-up-drug-sales.html?_r=1
9. http://blogs.wsj.com/corporate-intelligence/2014/03/13/the-pharmaceutical-sales-rep-lives-to-fight-another-day/
10. http://www.theatlantic.com/magazine/archive/2006/04/the-drug-pushers/304714
11. https://www.opensecrets.org/lobby/top.php?showYear=a&indexType=i
12. http://arstechnica.com/tech-policy/2014/05/how-the-patent-trolls-won-in-congress/
13. https://www.opensecrets.org/politicians/industries.php?type=C&cid=N00009922&newMem=N&cycle=2014
14. http://www.cbsnews.com/news/under-the-influence/3/
15. http://www.cbsnews.com/news/fda-harsh-criticism-from-within/
16. http://www.nytimes.com/2007/10/12/us/12gift.html
17. http://blogs.wsj.com/pharmalot/2015/02/20/what-money-most-docs-havent-visited-the-open-payments-database/
18. http://www.consumerreports.org/cro/health/suprising-way-big-pharma-keeps-drug-prices-high?kwp_0=53174&utm_keyword=creative1
19. http://cepr.net/documents/efficient_medicare_2006_01.pdf
20. https://bangordailynews.com/2016/01/18/opinion/contributors/maine-nurses-oppose-pacific-trade-pacts-gifts-to-billionaire-drug-companies/?ref=mostReadBoxOpinion


